Abstract
Il Censimento del Gruppo di Progetto di Dialisi Peritoneale (DP) della Società Italiana di Nefrologia ha seguito l’evoluzione della DP per 20 anni, fino all’ultima edizione del 2024 a cui, per la prima volta, ha fatto seguito un audit telematico, condotto tra il 28/04/2025 ed il 09/05/2025.
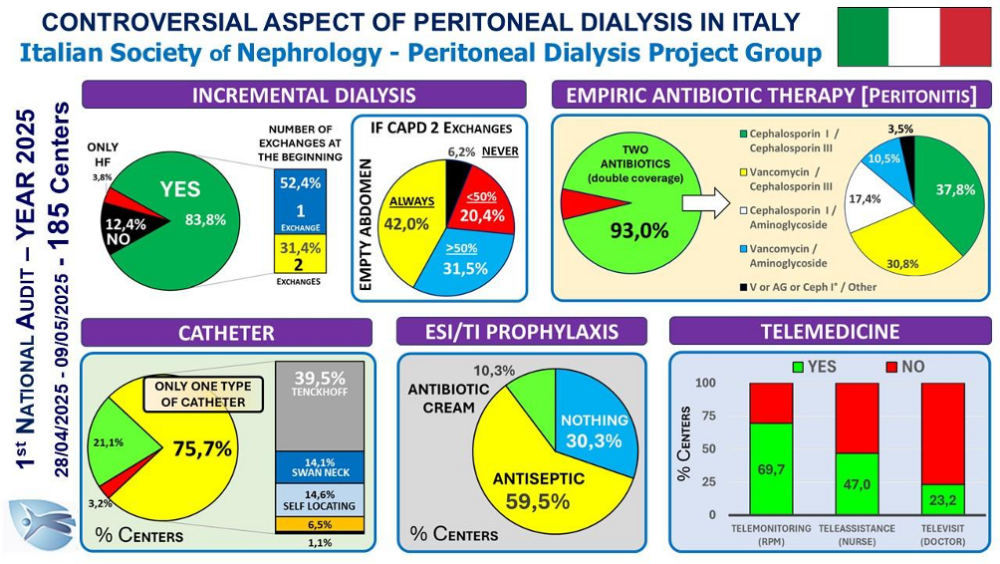
L’Audit, a cui hanno aderito 185 Centri dei 228 Centri che hanno utilizzato la DP nel 2024, ha indagato alcuni argomenti poco esplorati o controversi: 1) prescrizione dialitica incrementale in CAPD; 2) terapia empirica delle peritoniti; 3) profilassi delle exit site infections (ESI); 4) tipologia di catetere peritoneale utilizzato; 5) utilizzo della telemedicina.
Per la prescrizione incrementale in CAPD non vi è accordo. La maggior parte inizia con un solo scambio. In caso di due scambi la maggior parte mantiene l’addome vuoto per parte del giorno. Tuttavia, dei 162 Centri che utilizzano la DP incrementale, il 37,4% inizia solo a partire da 2 scambi ed il 26,5% nella CAPD a due scambi mantiene l’addome sempre pieno. Discreta variabilità è stata osservata anche per il tipo di catetere utilizzato anche se il 75,7% dei Centri ne utilizza un solo tipo nel proprio Centro.
La quasi totalità dei Centri segue quanto raccomandato dalla ISPD per la terapia empirica delle peritoniti e la profilassi delle ESI/TI, la cui validità è confermata dal costante calo del drop out per peritonite registrato negli ultimi 20 anni.
Infine, il dato sulla Telemedicina dimostra come questo strumento sia sempre più diffuso: il Telemonitoraggio risulta la modalità più utilizzata, mentre la Teleassistenza e soprattutto la Televisita, pur essendo molto utili nella DP assistita, sono impiegate solo da una minoranza di Centri.
Parole chiave: Dialisi Peritoneale, Dialisi Incrementale, Peritoniti, Exit Site Infections, Catetere peritoneale, Telemedicina